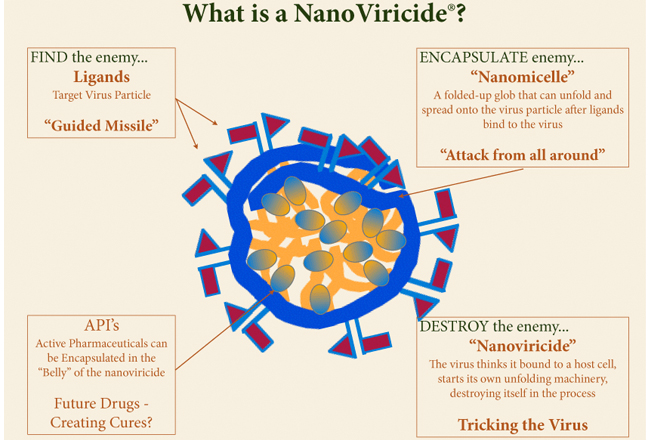
 Shelton pharmaceutical company NanoViricides has announced that its first drug candidate, NV-HHV-101, is on track with required preclinical GLP Safety and Toxicology studies moving toward human clinical trials.
Shelton pharmaceutical company NanoViricides has announced that its first drug candidate, NV-HHV-101, is on track with required preclinical GLP Safety and Toxicology studies moving toward human clinical trials.
NV-HHV-101 has been found to be safe and well tolerated in the clinical observation portion of the GLP Safety/Toxicology study of the drug as a dermal treatment for the shingles virus.
The first of the GLP studies was conducted using minipigs, which received twice daily skin treatment for 28 days, at different dosage levels. The animals were evaluated daily for general signs of toxicity including body weight, detailed clinical physical observations as well as the specific evaluation of the skin treatment areas. Topical treatment of the skin at all doses was well tolerated in all animals and all measured parameters remained within normal range in the study.
Additional studies required for the Safety and Toxicology datasets for filing an investigational new drug application are in progress.
NanoViricides is also testing the same drug for the treatment of HSV-1 cold sores and HSV-2 genital herpes. The market size for the company”™s immediate target drugs in its HerpeCide program is variously estimated into billions to tens of billions of dollars, according to NanoViricides.















